
Main Function
Targeting the process chain of technical service, R&D, pilot test service, achievement transformation, project incubation, industrialization, focus on the activities of transformation and incubation of scientific and technological achievements.
Quality control system
in accordance with CNAS (ISO/IEC17025), GMP & GLP requirements, the Institute has established a comprehensive system to achieve quality management throughout the whole process. System covers all factors and activities affecting the test result, including document management, organization structure, personnel management, instruments and equipment, sampling, analytical methods, quality assurance elements, and other standardized quality management systems.
The laboratory obtained the accreditation of CNAs in March 2017, In October, passed the qualification certificate of China Metrology Certification (CMA) laboratory.
In order to meet the requirements of data reliability in the whole process of drug research and development and testing, in 2018, the research institute upgraded the workstation system of all instruments according to the requirements of China GMP 2010 appendix computerization system, EU GMP appendix 11 computerization system, 21 CFR Part II electronic records electronic signatures, so as to ensure that users with corresponding authority can use and operate the system. Maintain and ensure all inspection activities can be audited and tracked.
Research and service system
The functional laboratory of the research institute covers an area of 10000 square meters, with more than 900 large and medium-sized instruments and equipment, with a total investment of 80 million RMB. It basically covers chemical drugs, biotechnology products, natural drugs, three categories of drugs, forming a preclinical research chain from source discovery to pilot test.
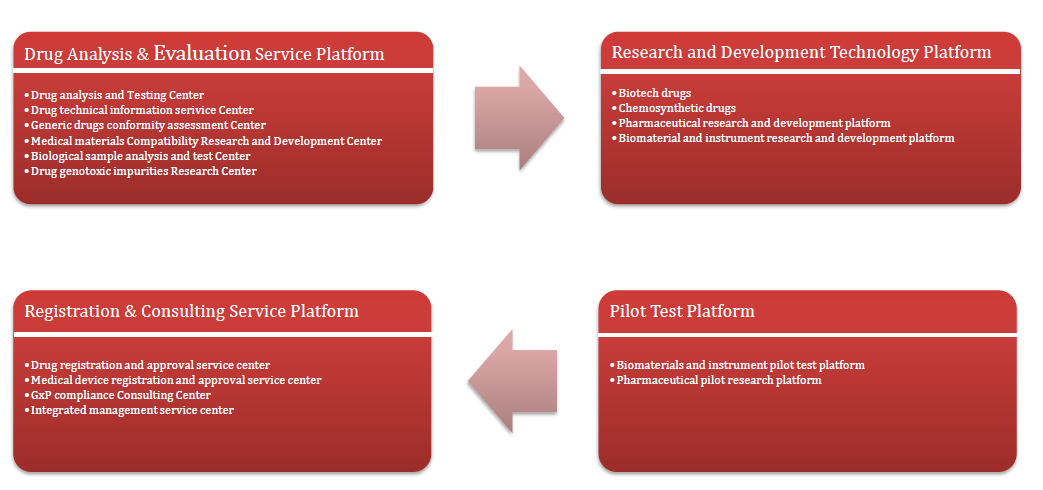
Technology Platform System
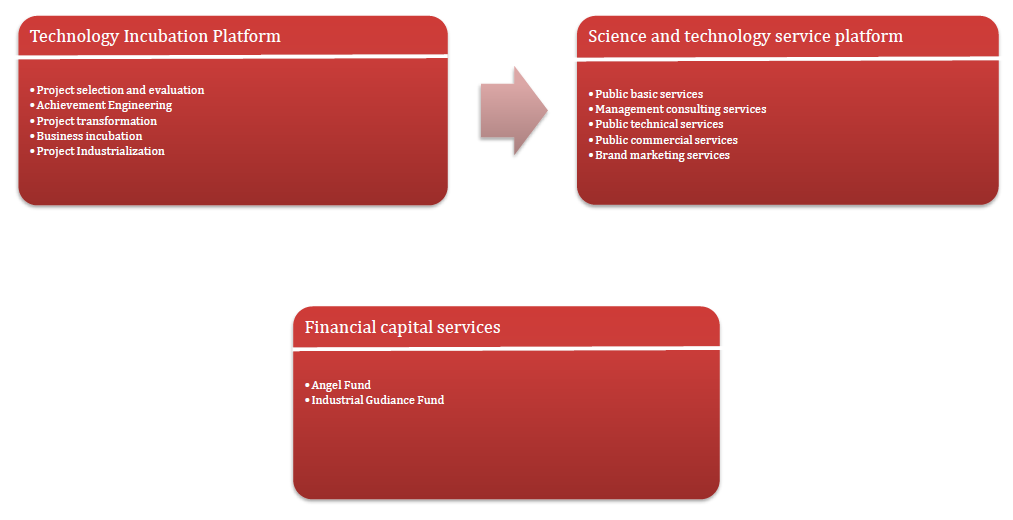
Incubation Service System
Cooperation Customers
